The approach was commonly referred to as VSEPR. Ronald Gillespie and Ronald Nyholm then developed the model into their theory published in 1957 they are considered the developers of the VSEPR theory. 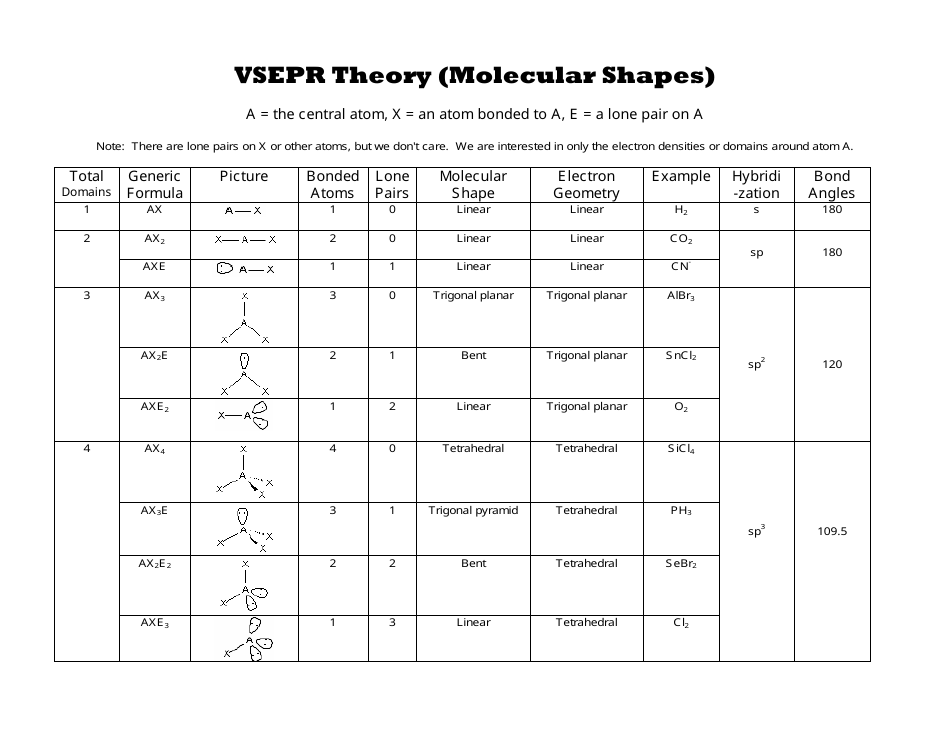
The model was proposed by Nevil Sidgwick and Herbert Powell in 1940. How to Determine Molecular Geometry YouTube: This video describes one method for quickly finding the major geometrical shapes for simple molecules. VSEPR is an acronym that stands for valence shell electron pair repulsion. 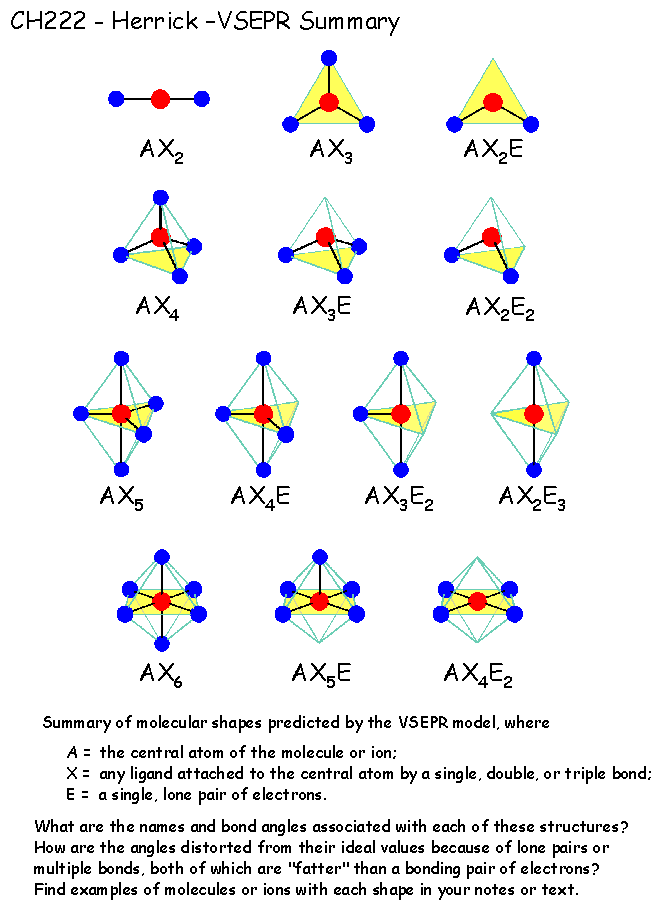
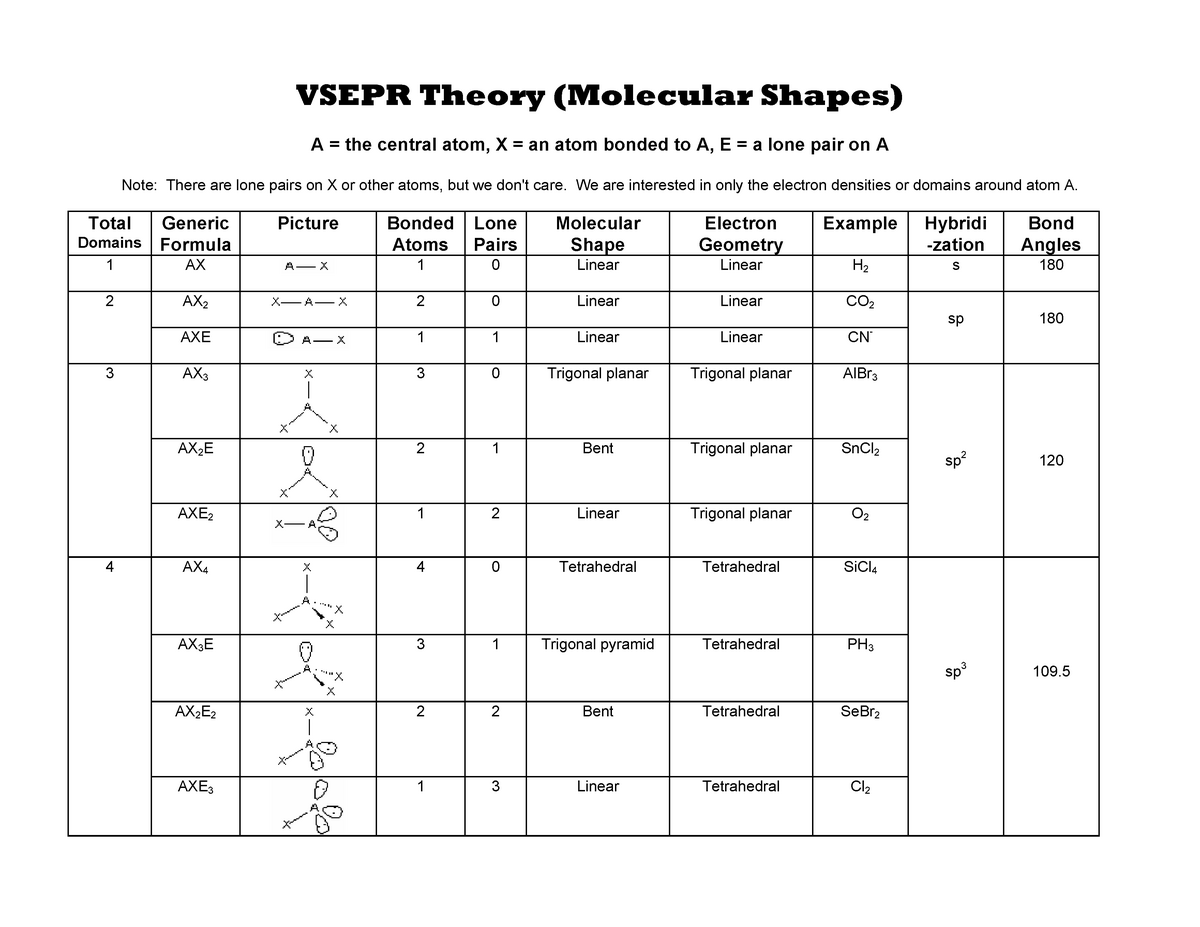
The bond angle, 115˚, slightly smaller than the trigonal-planar angle of 120˚, has been measured in the laboratory. VSEPR table of molecular geometries: The bonded angles in the table are ideal angles from the simple VSEPR theory the actual angle for the example given is in the following column. 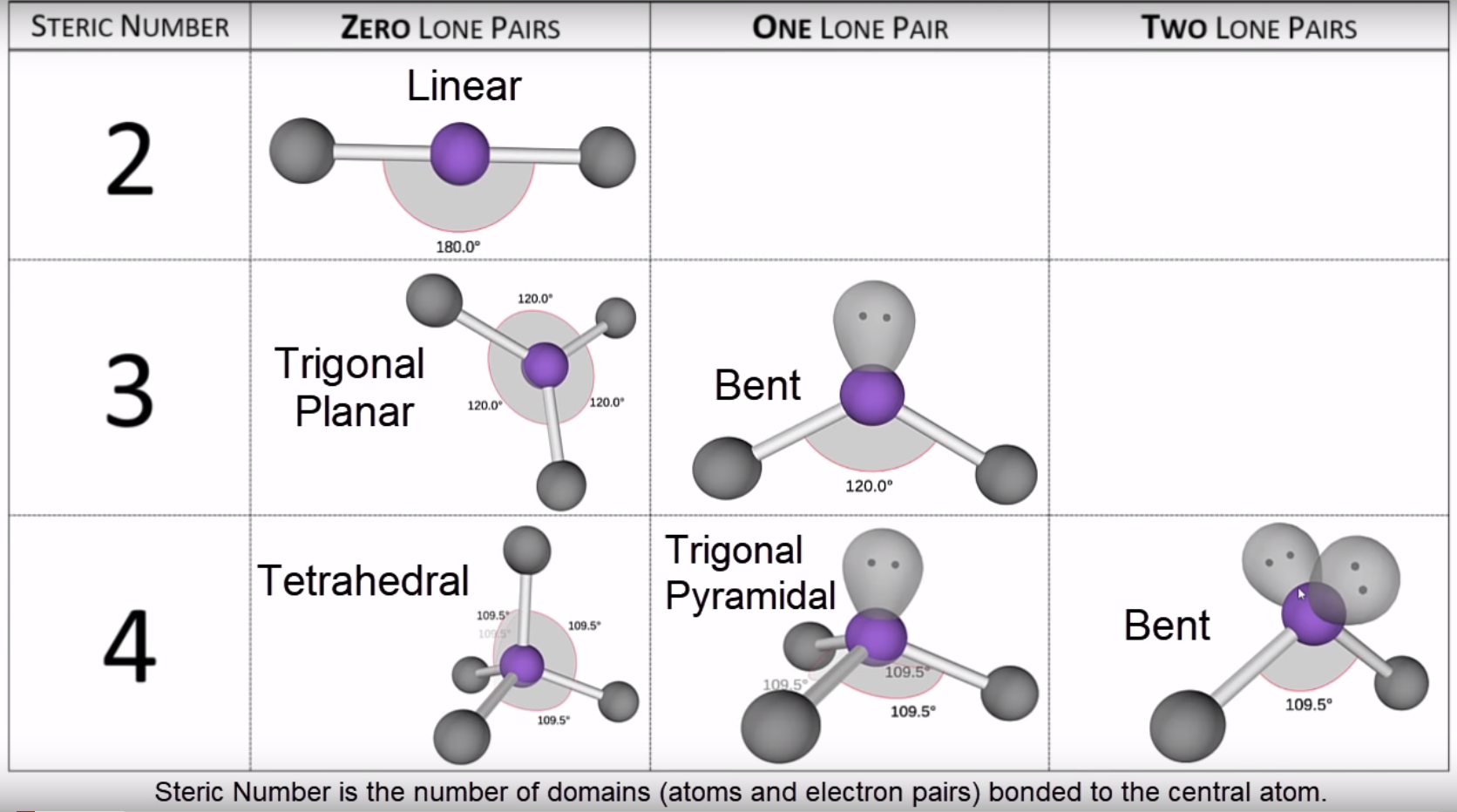
Which yields an overall structure that looks like this. The optimal structure is a resonance between these two structures In the single-double N–O arrangement, N has eight electrons, and the formal charges are lower, while still producing our overall -1 charge. The VSEPR model is not a theory it does not attempt to explain observations. First, N only has 6 valence electrons, and second, the formal charges might be reduced in another arrangement. The VSEPR model can predict the structure of nearly any molecule or polyatomic ion in which the central atom is a nonmetal, as well as the structures of many molecules and polyatomic ions with a central metal atom. The VSEPR theory is based on the assumption that the molecule will take shape such that electronic repulsion in the valence shell of that atom is. ISBN: 0 19 855694 2 (paperback) and 0 19 855695 0 (hardback) If you want other information on VSEPR try searching on VSEPR at the Sheffield Chemdex or try this VSEPR tutorial by John Nash at Purdue University, USA. The theory was first presented by Sidgwick and Powell in 1940. Recall that formal charge is (number of valence electrons) - (number of bonds) - (number of electrons in lone pairs).īecause there are two oxygens, our negative charge is accounted for here. Valence Shell Electron Pair Repulsion Theory ( VSEPR) is a molecular model to predict the geometry of the atoms making up a molecule where the electrostatic forces between a molecules valence electrons are minimized around a central atom. The VSEPR theory is used to predict the shape of the molecules from the electron pairs that surround the central atoms of the molecule. The formal charges on the nitrogen and the two (equivalent) oxygens are shown. Multiple bonds (double or triple bond) are regarded as one electron group for VSEPR purpose. Geometries predicted using VSEPR theory (bonded groups only).Solution: The geometries of these two ions are related, but there are subtle differences that we can understand by looking at Lewis structures, formal charges and VSEPR theory.Ĭonsider one possible bonding arrangement of NO 2 -, with single N–O bonds. For VSEPR purpose, the terms shape and geometry are interchangeable electron pair and electron group are also interchangeable.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
